Abstract
-
Purpose
This study aimed to explore the effects of epigallocatechin gallate (EGCG) in critically ill patients using a mouse model.
-
Methods
C57BL/6 mice were divided into control and EGCG groups (n=8 per group). The EGCG group received a 0.1% EGCG solution for 2 weeks, after which the mice were intraperitoneally injected with a lethal dose of lipopolysaccharide to induce acute endotoxic shock. Indirect calorimetry was performed for 24 hours. Changes in body weight, epididymal fat weight, and survival were measured, together with serum lipid levels, interleukin-6 (IL-6), and superoxide dismutase (SOD) concentrations. The expression of peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) was determined using quantitative real-time polymerase chain reaction, and its serum concentration was subsequently measured.
-
Results
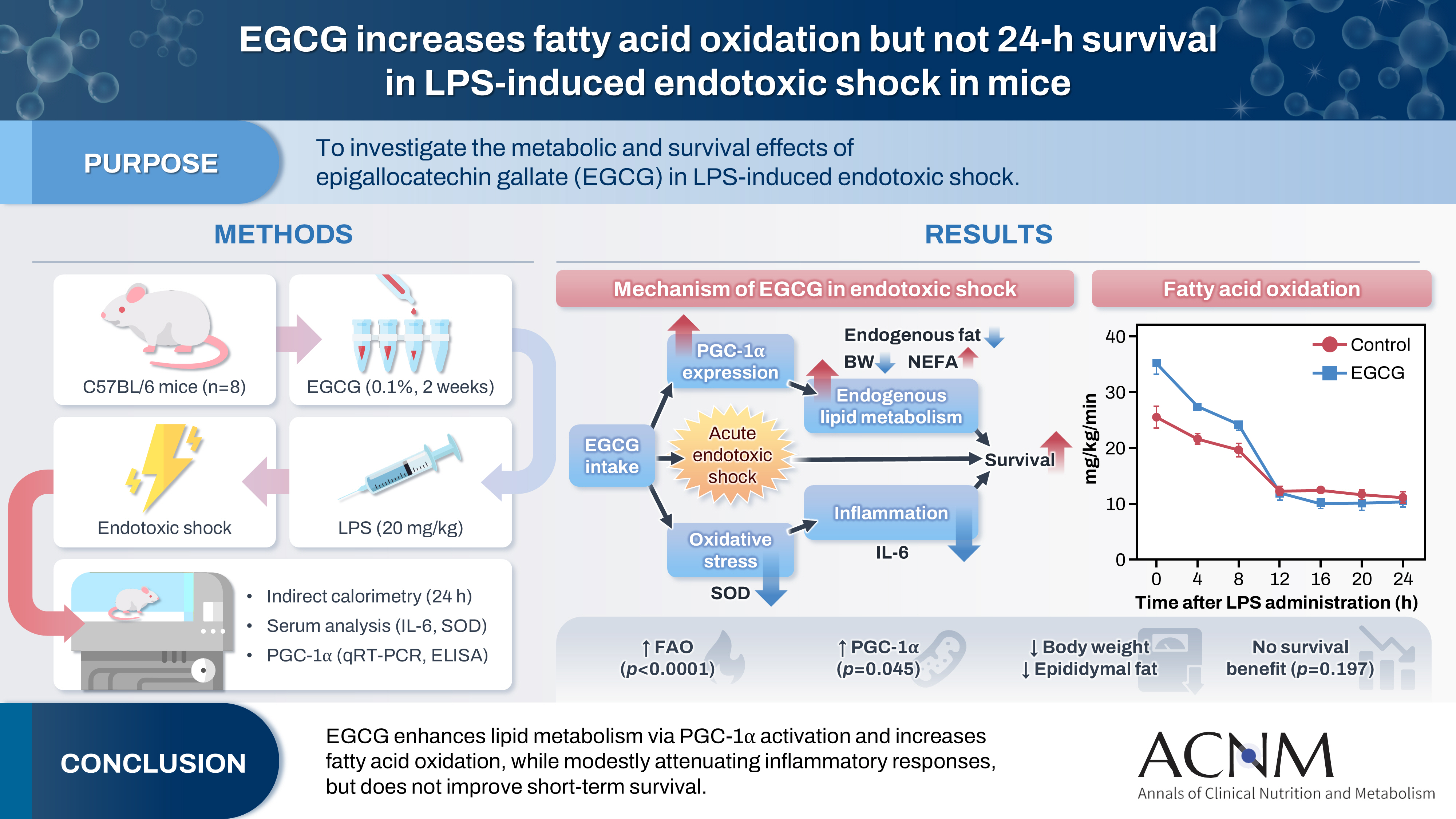
Indirect calorimetry showed a significant increase in fatty acid oxidation (P<0.0001) in the EGCG group, along with significant decreases in body weight and epididymal fat weight (P<0.01 and P<0.05, respectively). Survival did not differ significantly between groups (P=0.197). Serum lipid levels, IL-6, and SOD showed numerical differences, although these differences were not statistically significant. Furthermore, hepatic PGC-1α expression showed a tendency toward upregulation, and serum PGC-1α levels were significantly higher (P<0.05).
-
Conclusion
EGCG stimulates endogenous lipid metabolism through PGC-1α activation and may suppress inflammatory responses; therefore, it may represent a potentially useful nutrient for acute nutritional therapy.
-
Keywords: Critical illness; Epigallocatechin gallate; Fatty acids; Lipid metabolism; Survival
Graphical abstract
Introduction
Background
The importance of acute nutritional therapy for critically ill patients who have experienced severe trauma or sepsis is widely recognized, and various clinical guidelines have been developed to support evidence-based practice [
1,
2]. Among these recommendations, the use of indirect calorimetry to estimate energy expenditure is recommended with a GRADE 2B level of evidence in the latest Japanese guidelines [
3]. We have previously shown that metabolism shifts from carbohydrate-dominant to lipid-dominant metabolism during physiological stress and that therapeutic interventions targeting lipid metabolism may be effective [
4]. Specifically, peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), which plays a critical role in mitochondrial biogenesis and fatty acid β-oxidation [
5], is activated by interventions such as low-intensity exercise and neuromuscular electrical stimulation, leading to enhanced lipid metabolism and improved survival outcomes [
6,
7]. However, evidence regarding effective nutrients for acute nutritional therapy in critically ill patients remains limited. Although n-3 fatty acids and glutamine were previously considered potential immunomodulatory nutrients, large-scale clinical trials have reported negative results, and their use is no longer recommended [
8,
9]. Conversely, hydroxymethylbutyrate has shown some promise as a pharmaconutrient, particularly in enhancing recovery from exercise-induced muscle damage [
10].
Tea catechins are water-soluble polyphenols with a flavanol structure that are abundant in green tea, which is widely consumed in Japan and other Asian countries. Tea leaves contain approximately 30%–42% catechins by dry weight, with epigallocatechin gallate (EGCG) accounting for 50%–70% of the total catechin content. EGCG has been reported to exert antioxidant, anti-inflammatory, anticancer, and antibacterial effects and has also been associated with weight loss and anti-aging effects. This broad biological activity profile suggests potential utility not only in the management of chronic conditions but also in acute care settings, where inflammation and metabolic dysregulation are prominent. However, its effects in humans, particularly under acute stress conditions, remain unclear [
11]. Several studies have suggested that EGCG can activate PGC-1α [
12,
13]; however, its effects in conditions such as sepsis have not been thoroughly investigated. In addition, the potential of EGCG to influence mitochondrial function, modulate immune responses, and enhance metabolic flexibility makes it a promising candidate for therapeutic investigation in critical care settings.
This study aimed to investigate the potential role of EGCG in acute nutritional therapy for critically ill patients, with a particular focus on its effects on metabolic regulation and clinical outcomes under conditions of physiological stress.
Methods
Ethics statement
This study adhered to the ethical guidelines of the Aichi Medical University Animal Experimentation Committee (approval number: 2024-38) and complied fully with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Study design
EGCG feeding protocol
Eleven-week-old male C57BL/6 mice (Japan SLC) were used in this study. According to company data, the mice were produced under controlled conditions based on established production management standards, and no differences in body weight or biochemical characteristics were observed between individuals. The animals were acclimated in individual cages under a 12-hour light–dark cycle (light phase: 07:30–19:30 hours; dark phase: 19:30–07:30 hours) at a constant temperature (22±1 °C). Standard laboratory chow (Oriental Yeast) was provided
ad libitum. All animals were monitored daily for general health, food and water intake, and activity levels to ensure consistent baseline conditions before the intervention. Environmental enrichment was provided to minimize stress and maintain physiological stability throughout the experimental period. On day 1, the mice were randomly assigned to control or EGCG groups (n=8 per group). Tap water or a 0.1% EGCG solution was provided
ad libitum for 2 weeks. This concentration was calculated based on a report indicating that the non-toxic dose in mice is 500–750 mg/kg/day [
14] and that the average daily drinking volume in mice is approximately 5 mL/day [
15].
Body weight, epididymal fat weight, and serum lipid levels
Changes in body weight were measured after 2 weeks of EGCG intake. The mice were then sacrificed and dissected, and epididymal fat weight was measured. Liver and plasma samples were collected for analysis. To evaluate serum lipid levels, the concentration of non-esterified fatty acids (NEFA) was measured using a Wako spectrophotometry kit according to the manufacturer’s instructions.
Serum inflammatory cytokine and oxidative stress marker levels
To evaluate serum inflammatory cytokine and oxidative stress markers, plasma concentrations of interleukin-6 (IL-6) and superoxide dismutase (SOD) were measured using Quantikine enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems) according to the manufacturer’s instructions.
Liver and serum PGC-1α levels
Quantitative real-time polymerase chain reaction was performed to evaluate PGC-1α messenger RNA (mRNA) expression in the liver. Total RNA was extracted from liver tissue using TriPure Isolation Reagent (Roche Diagnostics) and reverse-transcribed into complementary DNA. Real-time polymerase chain reaction was performed using the TaqMan probe method with a LightCycler system (Roche Diagnostics). Hypoxanthine-guanine phosphoribosyltransferase served as the internal control. The primer sequences used were as follows: pgc1a, TGTGGAACTCTCTGGAACTGC (forward) and AGGGTTATCTTGGTTGGCTTTA (reverse); and hprt, TCCTCCTCAGACCGCTTTT (forward) and CCTGGTTCATCATCGCTAATC (reverse). Serum PGC-1α concentration was also measured using a Quantikine ELISA kit according to the manufacturer’s instructions.
Acute endotoxic shock mouse model
After 2 weeks of EGCG or tap-water intake, mice in both groups were intraperitoneally injected with a lethal dose of lipopolysaccharide (LPS) (20 mg/kg; Escherichia coli O55:B5, L-2880; Sigma-Aldrich) diluted in normal saline (10 mL/kg) to induce acute endotoxic shock. Food was withheld during the 24-hour metabolic assessment, whereas water was available ad libitum.
Indirect calorimetry and survival probability
Nutritional metabolism was assessed using indirect calorimetry for 24 hours after LPS administration using a mass spectrometer for respiratory gas analysis and a bioprocess monitoring system (ARCO-2000; Arco System Inc.). The system operated under controlled conditions of constant temperature (25±2 °C) and humidity (60%±10%). Data were collected every 10 minutes, and fatty acid oxidation (FAO) was calculated using the Frayn formula [
16]: FAO=1.67×VO
2–1.67×VCO
2.
Survival was monitored for 24 hours after LPS administration.
Statistical analysis
All values are presented as mean±standard error of the mean. Data between the two groups were analyzed using the Mann-Whitney U test. FAO data were analyzed using two-way analysis of variance with Sidak’s post hoc test. Survival data were analyzed using the Kaplan-Meier method with the log-rank test. Statistical analyses were performed using GraphPad Prism 9 software (GraphPad Inc.), and statistical significance was defined as P<0.05.
Results
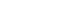
Body weight, epididymal fat weight, and serum lipid level
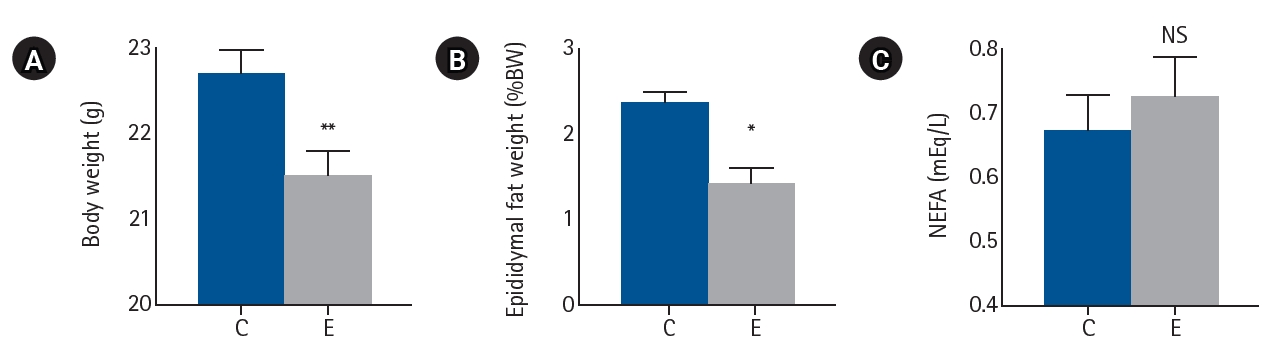
The mean EGCG intake was 193.5±4.5 mg/kg/day, which was well below the reported toxic dose. Body weight and epididymal fat weight were significantly lower in the EGCG group than in the control group (P=0.004 and P=0.029, respectively). Serum NEFA levels did not significantly differ between the groups (P=0.64). These findings suggest enhanced lipid utilization in the EGCG group (
Fig. 1).
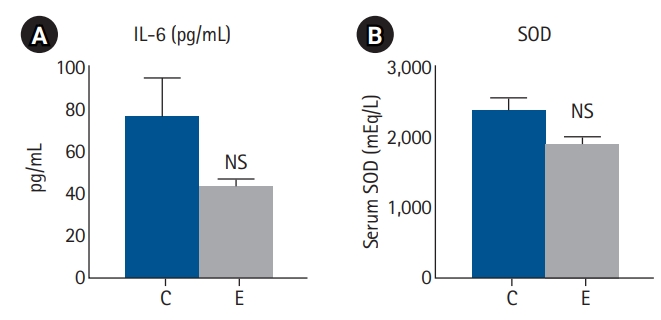
Serum IL-6 levels tended to be lower in the EGCG group than in the control group (P=0.098). Serum SOD levels also tended to be lower in the EGCG group (P=0.114). These findings suggest that EGCG may exert mild anti-inflammatory and antioxidant effects. The reductions in IL-6 and SOD levels, although not statistically significant, were consistent with previous reports describing the immunomodulatory properties of EGCG (
Fig. 2).
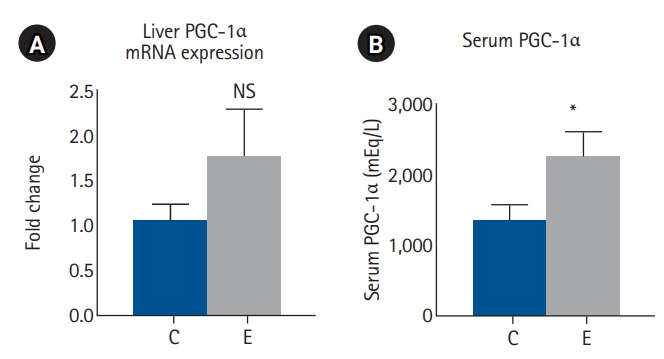
Compared with the control group, the EGCG group showed a higher hepatic PGC-1α mRNA expression (P=0.99) and significantly higher serum PGC-1α levels (P=0.045). These findings are consistent with the hypothesis that EGCG activates PGC-1α signaling pathways, potentially enhancing mitochondrial biogenesis and FAO (
Fig. 3).
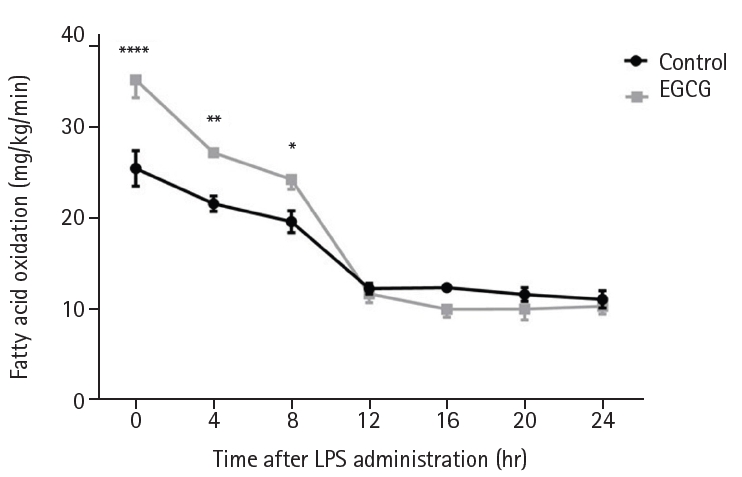
Indirect calorimetry showed that FAO was significantly higher in the EGCG group than in the control group (P=0.0001, P=0.006, and P=0.031 at 0, 4, and 8 hours after LPS administration, respectively) (
Fig. 4).
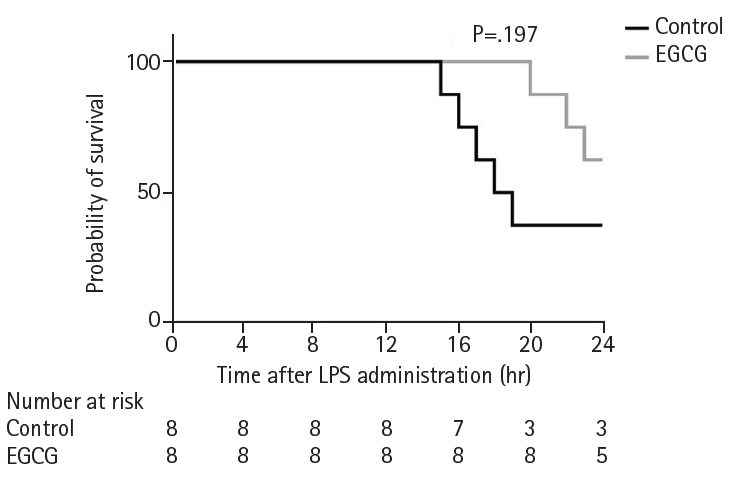
Survival did not differ significantly between the groups (P=0.197) (
Fig. 5).
Discussion
Key results
In this study, we investigated the effects of EGCG in a mouse model intended to reflect acute critical illness.
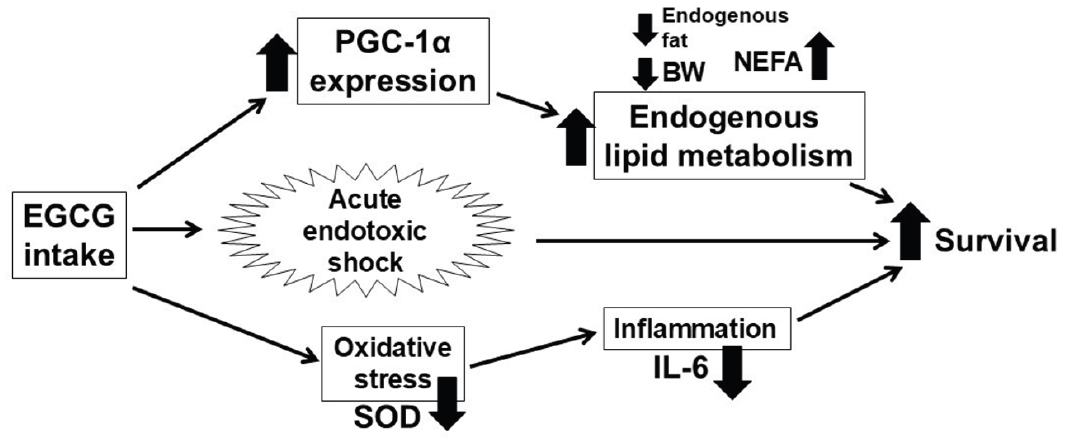
Fig. 6 illustrates the proposed mechanism underlying the effects of EGCG observed in this study. EGCG intake increased PGC-1α levels and endogenous lipid metabolism and suppressed oxidative stress and inflammation, but it did not significantly improve survival.
These findings are consistent with the hypothesis that EGCG may act as a metabolic modulator during acute physiological stress. Indirect calorimetry is useful for assessing individual metabolic changes under stress conditions and for applying these data to acute nutritional therapy; accordingly, studies evaluating its clinical utility are increasing [
17]. This method may be important for precision medicine and personalized nutrition therapy [
18,
19], both of which have received increasing attention in intensive care. By tailoring nutritional interventions to a patient’s real-time metabolic status, clinicians may be able to optimize recovery and reduce complications.
PGC-1α plays an important role in energy metabolism and is thought to improve lipid metabolism. Exercise, fasting, and cold exposure activate PGC-1α [
20]. We have previously shown that low-intensity exercise during the acute phase affects PGC-1α and lipid metabolism and improves survival [
9]. In addition, specific neuromuscular electrical stimulation conditions increase PGC-1α and lipid metabolism, suppress inflammation, and improve survival [
10]. These interventions may be applicable to critically ill patients with hemodynamic instability. However, nutritional components with similar effects have not been adequately investigated. This gap underscores the importance of exploring bioactive nutrients such as EGCG, which may offer similar benefits without requiring physical exertion, which is often not feasible in critically ill patients.
Tea catechins are distinctive bioactive compounds found in green tea, and EGCG in particular is believed to have several beneficial effects, including anti-inflammatory, antioxidant, and fat-reducing effects. EGCG has been reported to reduce body fat after long-term intake in humans [
21] and to exert antioxidant and antibacterial effects [
22,
23]. On this basis, we considered it a potentially useful nutrient for acute nutritional therapy in critically ill patients. Its natural origin and widespread dietary use make green tea an attractive candidate for integration into clinical nutritional protocols, particularly in regions where green tea consumption is culturally prevalent.
The findings of this study suggest that EGCG may exert metabolic regulatory effects in acute endotoxic shock by increasing endogenous lipid metabolism through PGC-1α activation and by suppressing inflammatory responses. Here, “metabolic resuscitation” refers to improvement in outcomes through recovery from metabolic dysfunction during severe stress, such as sepsis. EGCG may be a useful nutrient during early enteral nutrition in critically ill patients. This approach may be particularly helpful for patients who cannot tolerate physical interventions such as exercise or neuromuscular electrical stimulation, because it may provide a noninvasive means of stimulating metabolic recovery and potentially enhancing the efficacy of early nutritional support, which is already known to be beneficial in critical care. Moreover, the compatibility of EGCG with enteral nutrition formulations makes it a practical candidate for incorporation into existing intensive care unit protocols.
Limitations
First, this study was conducted in a mouse model, and the optimal dose in humans remains unknown. In our mouse experiment, the administered intake was below the reported toxic dose; however, liver injury and other adverse effects have been reported in humans [
24]. Therefore, careful attention should be paid to the dose administered in humans. In addition, the pharmacokinetics and bioavailability of EGCG in critically ill patients may differ substantially from those in healthy individuals, necessitating careful clinical evaluation. Second, in this study, EGCG was administered before LPS administration, not after the onset of acute endotoxic shock. Forced administration of a fixed amount of EGCG to mice after LPS administration was not feasible because the procedure itself caused substantial stress and increased mortality. In the clinical setting, whether similar effects would occur when EGCG is administered after hospital arrival, that is, after disease onset, remains unclear. Finally, because of the short duration of the experiment, the effects on long-term outcomes remain unknown. Future studies should investigate the therapeutic window, optimal dosing strategies, and long-term safety and effects of EGCG in humans.
EGCG may be a useful nutrient for acute nutritional therapy in critically ill patients. Our findings suggest that EGCG may serve as a novel adjunctive therapy in critical care, particularly during the early phase of enteral nutrition. Further clinical studies are required to determine its effects in humans, including the optimal dose, timing, and delivery method, as well as its long-term safety and efficacy. If validated, EGCG could be incorporated into personalized nutritional strategies to support metabolic recovery and improve outcomes in critically ill patients.
Authors’ contribution
Conceptualization: RK, DO, TT. Formal analysis: TI, MMI, US, SM, RA. Investigation: TI, MMI, US, SM, RA. Supervision: EW. Writing–original draft: TI, RK, DO, TT. Writing–review & editing: all authors. All authors read and approved the final manuscript.
Conflict of interest
The authors of this manuscript have no conflicts of interest to disclose.
Funding
This study was supported by a Grant-in-Aid for Scientific Research (KAKENHI; Grant No. 24K19513) funded by the Japan Society for the Promotion of Science. The funder played no role in the design, data collection, data analysis, or reporting of this study.
Data availability
Contact the corresponding author for research data availability.
Acknowledgments
The authors thank the research assistants and other staff members at the Department of Emergency and Critical Care Medicine, Aichi Medical University for their assistance in this study.
Supplementary materials
None.
Fig. 1.Effects of EGCG on metabolic parameters. (A) Body weight, (B) epididymal fat weight, and (C) serum NEFA levels. Values are presented as mean±standard error of the mean (n=8 per group) and were analyzed using the Mann-Whitney U test. C, control group; E, epigallocatechin gallate (EGCG) group; NS, not significant; NEFA, non-esterified fatty acid. *P<0.05, **P<0.01 vs. C.
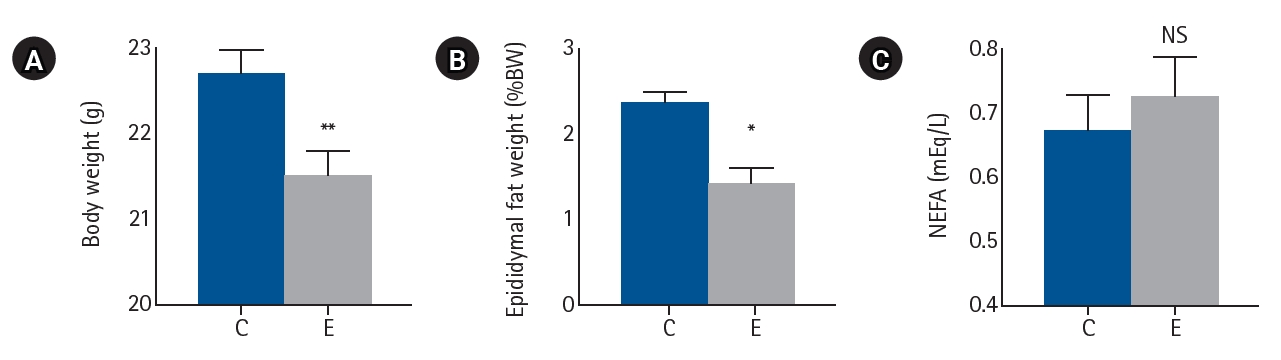
Fig. 2.Serum inflammatory response markers. (A) Serum IL-6 and (B) SOD levels. Values are presented as mean±standard error of the mean (n=8 per group for IL-6 and n=4 per group for SOD) and were analyzed using the Mann-Whitney U test. IL-6, interleukin-6; SOD, superoxide dismutase; C, control group; E, epigallocatechin gallate (EGCG) group; NS, not significant.
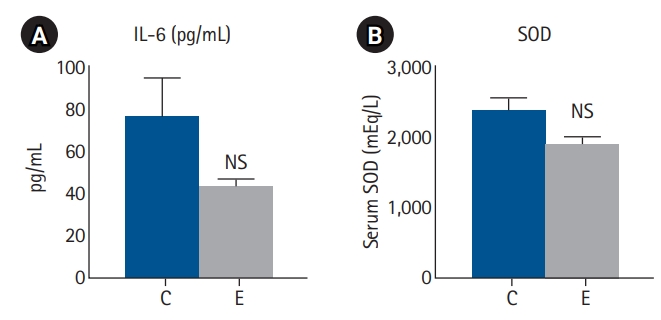
Fig. 3.PGC-1α levels in liver and serum. (A) Liver PGC-1α levels. (B) Serum PGC-1α levels. Values are presented as mean±standard error of the mean (n=8 per group) and were analyzed using the Mann-Whitney U test. PGC-1α, proliferator-activated receptor gamma coactivator 1-alpha; mRNA, messenger RNA; C, control group; E, epigallocatechin gallate (EGCG) group; NS, not significant. *P<0.05 vs. C.
Fig. 4.Indirect calorimetry. Values are presented as mean±standard error of the mean (n=8 per group). Fatty acid oxidation was analyzed using two-way analysis of variance with Sidak post hoc test. EGCG, epigallocatechin gallate; LPS, lipopolysaccharide. *P<0.05, **P<0.01, ****P<0.0001 vs. control.
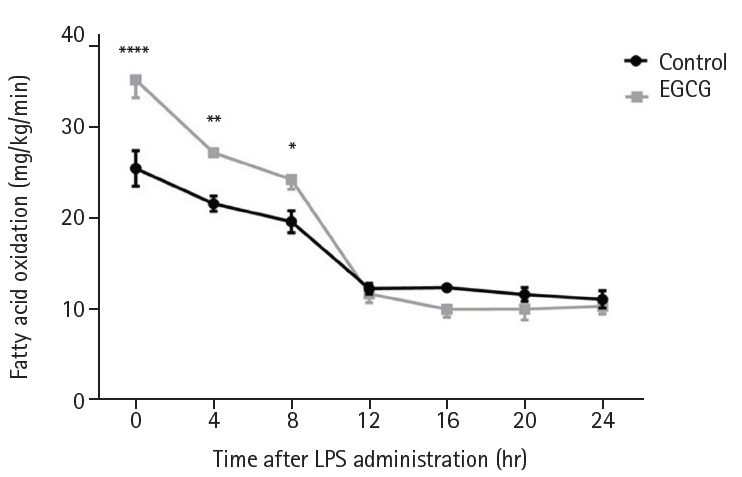
Fig. 5.Survival proportions. Survival proportions were analyzed using the Kaplan-Meier method and the log-rank test. EGCG, epigallocatechin gallate; LPS, lipopolysaccharide.
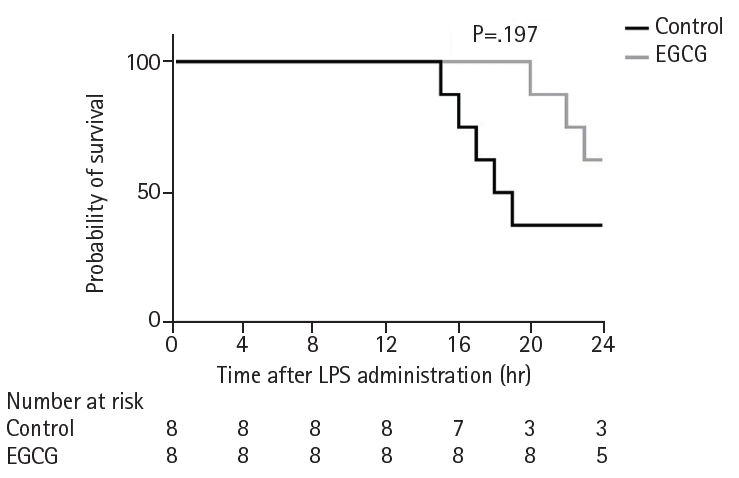
Fig. 6.Proposed mechanism of EGCG intake in acute endotoxic shock. EGCG, epigallocatechin gallate; PGC-1α, proliferator-activated receptor gamma coactivator 1-alpha; SOD, superoxide dismutase; BW, body weight; NEFA, non-esterified fatty acid; IL-6, interleukin-6.
References
- 1. McClave SA, Taylor BE, Martindale RG, Warren MM, Johnson DR, Braunschweig C, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically Ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). JPEN J Parenter Enteral Nutr 2016;40:159-211. ArticlePubMed
- 2. Singer P, Blaser AR, Berger MM, Calder PC, Casaer M, Hiesmayr M, et al. ESPEN practical and partially revised guideline: clinical nutrition in the intensive care unit. Clin Nutr 2023;42:1671-89. ArticlePubMed
- 3. Nakamura K, Yamamoto R, Higashibeppu N, Yoshida M, Tatsumi H, Shimizu Y, et al. The Japanese critical care nutrition guideline 2024. J Intensive Care 2025;13:18.ArticlePubMedPMC
- 4. Irahara T, Sato N, Otake K, Matsumura S, Inoue K, Ishihara K, et al. Alterations in energy substrate metabolism in mice with different degrees of sepsis. J Surg Res 2018;227:44-51. ArticlePubMed
- 5. Rius-Perez S, Torres-Cuevas I, Millan I, Ortega AL, Perez S. PGC-1α, inflammation, and oxidative stress: an integrative view in metabolism. Oxid Med Cell Longev 2020;2020:1452696.ArticlePubMedPMC
- 6. Irahara T, Sato N, Inoue K, Otake K, Ohtsuru S, Koike K, et al. Low-intensity exercise in the acute phase of lipopolysaccharide-induced sepsis improves lipid metabolism and survival in mice by stimulating PGC-1α expression. J Trauma Acute Care Surg 2016;80:933-40. ArticlePubMed
- 7. Irahara T, Sato N, Otake K, Murata S, Inoue K, Koike K, et al. Neuromuscular electrical stimulation improves energy substrate metabolism and survival in mice with acute endotoxic shock. Shock 2020;53:236-41. ArticlePubMedPMC
- 8. Rice TW, Wheeler AP, Thompson BT, deBoisblanc BP, Steingrub J, Rock P, et al. Enteral omega-3 fatty acid, gamma-linolenic acid, and antioxidant supplementation in acute lung injury. JAMA 2011;306:1574-81. ArticlePubMedPMC
- 9. van Zanten AR, Sztark F, Kaisers UX, Zielmann S, Felbinger TW, Sablotzki AR, et al. High-protein enteral nutrition enriched with immune-modulating nutrients vs standard high-protein enteral nutrition and nosocomial infections in the ICU: a randomized clinical trial. JAMA 2014;312:514-24. ArticlePubMed
- 10. Wilson JM, Fitschen PJ, Campbell B, Wilson GJ, Zanchi N, Taylor L, et al. International Society of Sports Nutrition Position Stand: beta-hydroxy-beta-methylbutyrate (HMB). J Int Soc Sports Nutr 2013;10:6.ArticlePubMedPMCPDF
- 11. Radeva-Ilieva M, Stoeva S, Hvarchanova N, Georgiev KD. Green tea: current knowledge and issues. Foods 2025;14:745.ArticlePubMedPMC
- 12. Ye Q, Ye L, Xu X, Huang B, Zhang X, Zhu Y, et al. Epigallocatechin-3-gallate suppresses 1-methyl-4-phenyl-pyridine-induced oxidative stress in PC12 cells via the SIRT1/PGC-1α signaling pathway. BMC Complement Altern Med 2012;12:82.ArticlePubMedPMCPDF
- 13. Lee MS, Lee S, Doo M, Kim Y. Green tea (-)-epigallotocatechin-3-gallate induces PGC-1α gene expression in HepG2 cells and 3T3-L1 adipocytes. Prev Nutr Food Sci 2016;21:62-7. ArticlePubMedPMC
- 14. Lambert JD, Kennett MJ, Sang S, Reuhl KR, Ju J, Yang CS, et al. Hepatotoxicity of high oral dose (-)-epigallocatechin-3-gallate in mice. Food Chem Toxicol 2010;48:409-16. ArticlePubMedPMC
- 15. National Research Council. Guide for the Care and Use of Laboratory Animals, 8th ed. National Academies Press; 2011.
- 16. Frayn KN. Calculation of substrate oxidation rates in vivo from gaseous exchange. J Appl Physiol Respir Environ Exerc Physiol 1983;55:628-34. ArticlePubMed
- 17. Duan JY, Zheng WH, Zhou H, Xu Y, Huang HB. Energy delivery guided by indirect calorimetry in critically ill patients: a systematic review and meta-analysis. Crit Care 2021;25:88.ArticlePDF
- 18. Buchman TG, Billiar TR, Elster E, Kirk AD, Rimawi RH, Vodovotz Y, et al. Precision medicine for critical illness and injury. Crit Care Med 2016;44:1635-8. ArticlePubMed
- 19. Wischmeyer PE, Bear DE, Berger MM, De Waele E, Gunst J, McClave SA, et al. Personalized nutrition therapy in critical care: 10 expert recommendations. Crit Care 2023;27:261.ArticlePubMedPMCPDF
- 20. Lin J, Handschin C, Spiegelman BM. Metabolic control through the PGC-1 family of transcription coactivators. Cell Metab 2005;1:361-70. ArticlePubMedPMC
- 21. Murase T, Nagasawa A, Suzuki J, Hase T, Tokimitsu I. Beneficial effects of tea catechins on diet-induced obesity: stimulation of lipid catabolism in the liver. Int J Obes Relat Metab Disord 2002;26:1459-64. ArticlePubMedPDF
- 22. Wheeler DS, Lahni PM, Hake PW, Denenberg AG, Wong HR, Snead C, et al. The green tea polyphenol epigallocatechin-3-gallate improves systemic hemodynamics and survival in rodent models of polymicrobial sepsis. Shock 2007;28:353-9. ArticlePubMed
- 23. Li W, Ashok M, Li J, Yang H, Sama AE, Wang H, et al. A major ingredient of green tea rescues mice from lethal sepsis partly by inhibiting HMGB1. PLoS One 2007;2:e1153.ArticlePubMedPMC
- 24. Mazzanti G, Menniti-Ippolito F, Moro PA, Cassetti F, Raschetti R, Santuccio C, et al. Hepatotoxicity from green tea: a review of the literature and two unpublished cases. Eur J Clin Pharmacol 2009;65:331-41. ArticlePubMedPDF

 , Ryusuke Katsuki1
, Ryusuke Katsuki1 , Dai Oishi1, Tsuguaki Terashima1, Md. Monirul Islam2
, Dai Oishi1, Tsuguaki Terashima1, Md. Monirul Islam2 , Umme Salma1
, Umme Salma1 , Shohag Majumder1
, Shohag Majumder1 , Ridwan Ahmed1
, Ridwan Ahmed1 , Eizo Watanabe1
, Eizo Watanabe1








 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN
 Cite
Cite