Scopus, KCI, KoreaMed

Articles
- Page Path
- HOME > Ann Clin Nutr Metab > Volume 18(1); 2026 > Article
- Review Effectiveness of perioperative rehabilitation and nutrition in esophageal cancer: a narrative review
-
Ryoma Haneda1
 , Yuki Sakai1, Kenichi Sekimori1, Tomohiro Murakami1, Eisuke Booka1, Tomohiro Matsumoto1, Hirotoshi Kikuchi1, Yoshihiro Hiramatsu1,2, Hiroya Takeuchi1
, Yuki Sakai1, Kenichi Sekimori1, Tomohiro Murakami1, Eisuke Booka1, Tomohiro Matsumoto1, Hirotoshi Kikuchi1, Yoshihiro Hiramatsu1,2, Hiroya Takeuchi1 -
Annals of Clinical Nutrition and Metabolism 2026;18(1):18-26.
DOI: https://doi.org/10.15747/ACNM.25.0021
Published online: March 30, 2026
1Department of Surgery, Hamamatsu University School of Medicine, Hamamatsu, Japan
2Department of Perioperative Functioning Care and Support, Hamamatsu University School of Medicine, Hamamatsu, Japan
- Corresponding author: Hiroya Takeuchi, email: takeuchi@hama-med.ac.jp
This article is a secondary publication of an article in Japanese published in the Japanese Journal of Surgical Metabolism and Nutrition, 2024;58(6):216-222. https://doi.org/10.11638/jssmn.58.6_216. The editors of both journals have granted permission for secondary publication.
This study was presented at the 60th Annual Meeting of the Japanese Society of Surgical Metabolism and Nutrition, held on July 7, 2023, in Tokyo, Japan.
© 2026 The Korean Society of Surgical Metabolism and Nutrition · The Korean Society for Parenteral and Enteral Nutrition · Asian Society of Surgical Metabolism and Nutrition
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 624 Views
- 15 Download
Abstract
-
Purpose Esophageal cancer surgery requires robust perioperative management to reduce its high rate of complications. This review evaluates the clinical utility of comprehensive exercise and nutritional therapy, with a specific focus on initiatives led by the multidisciplinary Hamamatsu Perioperative Care Team (HOPE), established at Hamamatsu University Hospital to optimize patient safety and postoperative recovery outcomes.
-
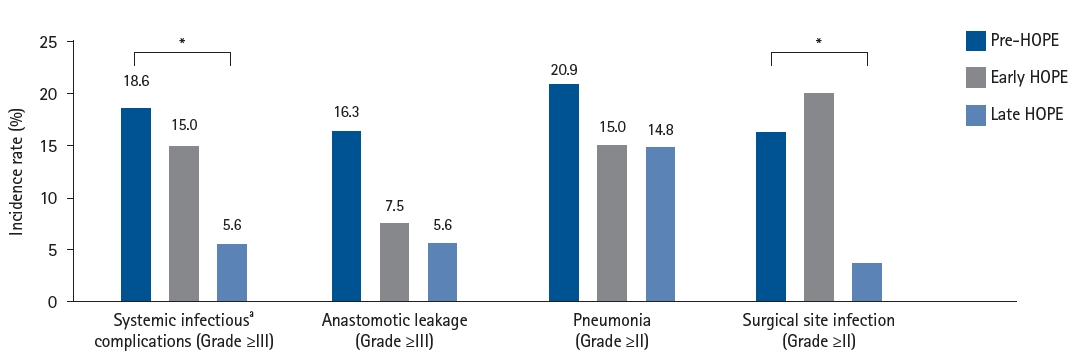
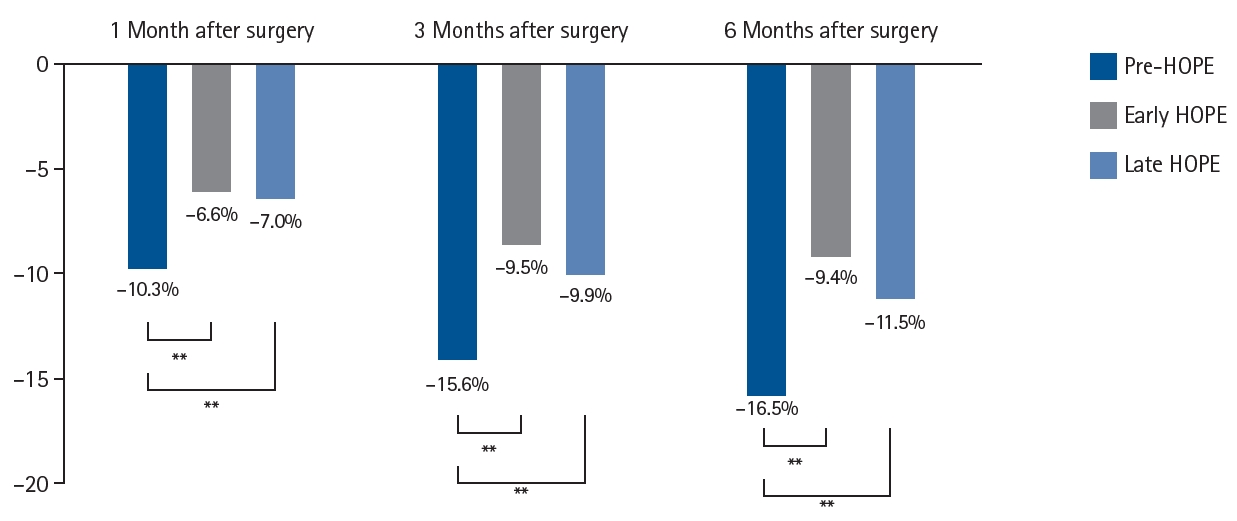
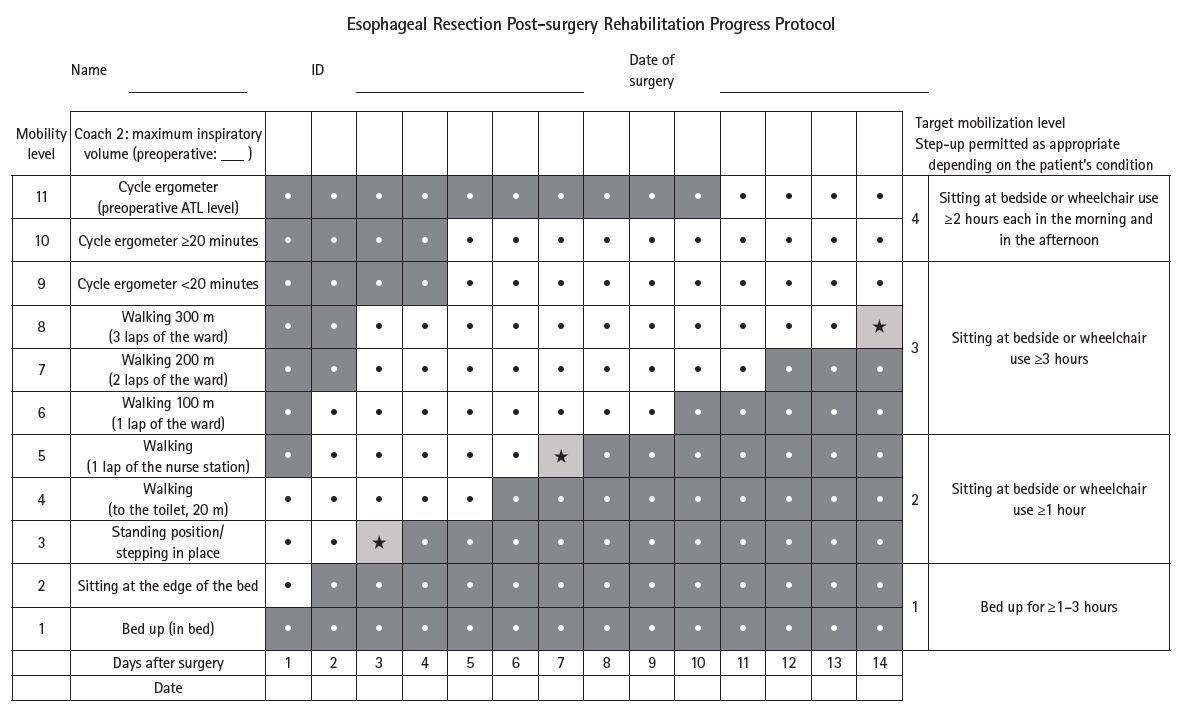
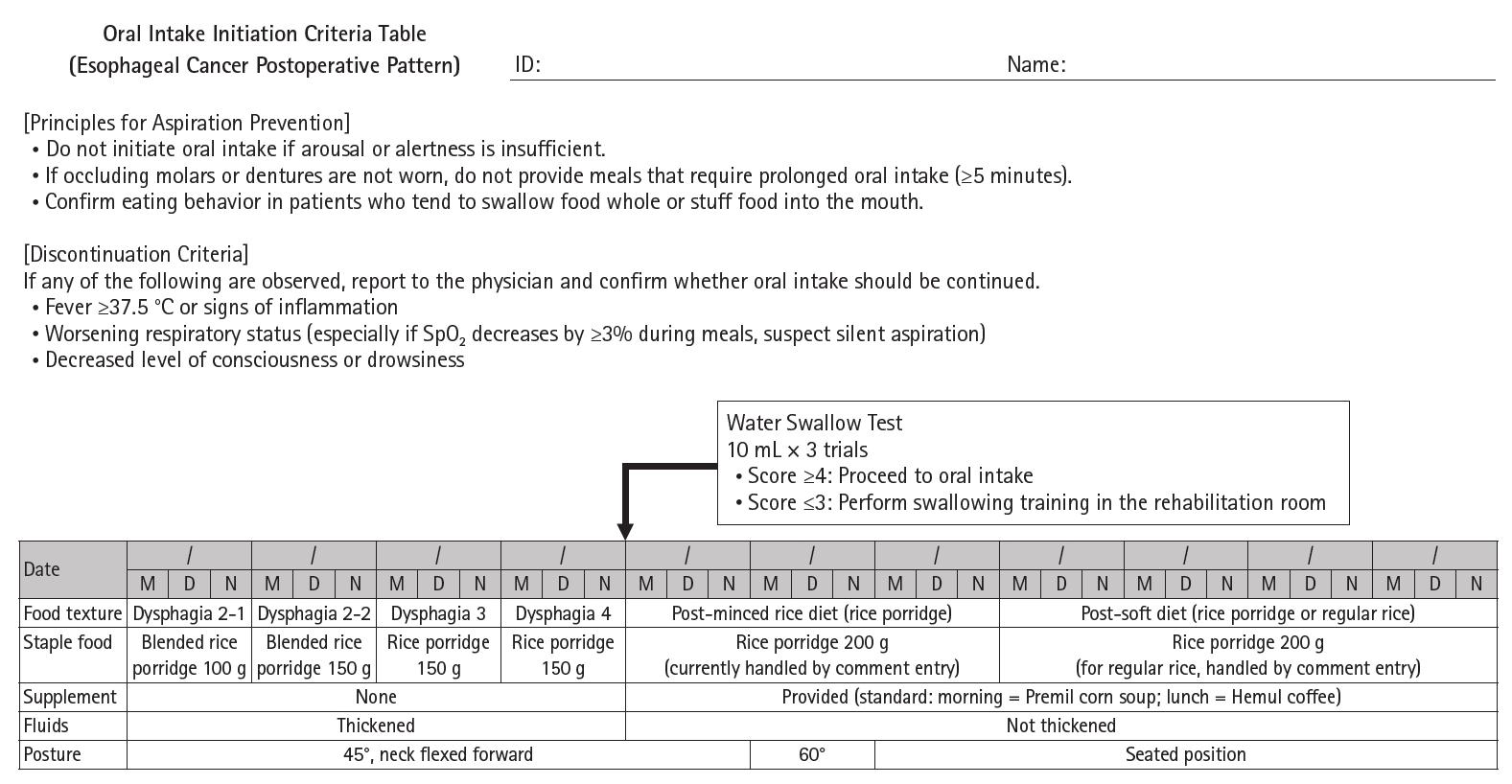
Current concept The HOPE protocol involves a diverse team, including surgeons, nurses, pharmacists, and dietitians, who collaboratively implement a multifaceted perioperative care bundle. These interventions include strict smoking cessation, intensive oral care, and immunonutrition incorporating n-3 fatty acids. A distinctive feature of this program is the integration of patient-reported treatment diaries and wearable fitness tracking devices (WFTs) to visualize daily activity, thereby helping to sustain patient motivation. Preoperative rehabilitation focuses on preventing sarcopenia through combined aerobic and resistance training, while postoperative care emphasizes immediate enteral nutrition and early mobilization. Retrospective analyses demonstrated that HOPE implementation reduced Clavien-Dindo grade III or higher infectious complications, pneumonia and surgical site infections, compared with historical controls. In addition, a propensity score–matched analysis showed that WFT users experienced lower pneumonia rates, shorter hospital stays, and better preservation of nutritional markers, including albumin and transthyretin, during the recovery phase compared with non-users.
-
Conclusion The HOPE strategy illustrates that combining standard nutritional support and exercise therapy with digital tools enables individualized rehabilitation. This integrated approach reduces morbidity and preserves function, strongly suggesting that incorporation of wearable technology into established surgical care protocols represents a strategy for improving long-term outcomes in high-risk cancer patients.
Introduction
Perioperative team medicine
Results and future issues of perioperative team medicine
Authors’ contribution
Conceptualization: HT. Data curation: YS, KS, TM (Tomohiro Murakami), EB, TM (Tomohiro Matsumoto), HK, YH. Methodology/formal analysis/validation: YS, KS, TM (Tomohiro Murakami), EB, TM (Tomohiro Matsumoto), HK, YH. Project administration: HT. Writing–original draft: RH. Writing–review & editing: RH, YS, KS, TM (Tomohiro Murakami), EB, TM (Tomohiro Matsumoto), HK, YH, HT. All authors read and approved the final manuscript.
Conflict of interest
The authors of this manuscript have no conflicts of interest to disclose.
Funding
None.
Data availability
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
None.
Supplementary materials
None.
| Perioperative management team | Author | Preoperative | Postoperative | Outcome |
|---|---|---|---|---|
| PERiO | Shirakawa et al. (2019) [3] | Smoking cessation guidance, physical ability assessment, exercise therapy, oral care | Physical ability assessment, respiratory physiotherapy, basic movement assistance, eating/swallowing rehabilitation | Fewer days to postoperative ambulation, shortened hospital stay, reduced adverse events during preoperative chemotherapy |
| Shirakawa et al. (2021) [4] | ||||
| PeriCan | Watanabe et al. (2016) [5] | Alcohol and smoking cessation guidance, oral care | Early mobilization, rehabilitation, swallowing function assessment, dietary guidance | Reduced pneumonia |
| Perioperative Center | Ochiai (2018) [6] | Patient orientation, oral care, medication adjustment, deep vein thrombosis prevention | Postoperative pain control, oral care | Shortened average hospital stay |
| PMT | Yamamoto and Sakakibara (2023) [7] | Smoking/alcohol cessation guidance, respiratory rehabilitation, nutritional guidance, oral care, skin protection, consultation for use of economic/social resources (post-discharge life support, nursing care, etc.) | Continued postoperative nursing, discharge support, utilization of social resources | Reduced surgery postponement, optimized hospital stays, prevention of complications |
| PMT | Yokoyama (2021) [8] | Physical and social background, patient/family acceptance of surgery, swallowing function assessment, risk of postoperative nausea/vomiting, preoperative pain management plan | Treatment course, evaluation of future PMT interventions, coordination with each committee | Identification of high-risk patients for swallowing disorders |
| Tobu Hospital Patient Support Center | Ishida (2023) [9] | Assessment of dietary intake, nutritional guidance, grip strength/body measurement, calculation of nutritional indices, body composition measurement, prehabilitation (aerobic exercise, resistance exercise, stretching, respiratory training), oral care | Nutritional management, rehabilitation | Increased walking distance, improved sit-and-reach, increased skeletal muscle mass |
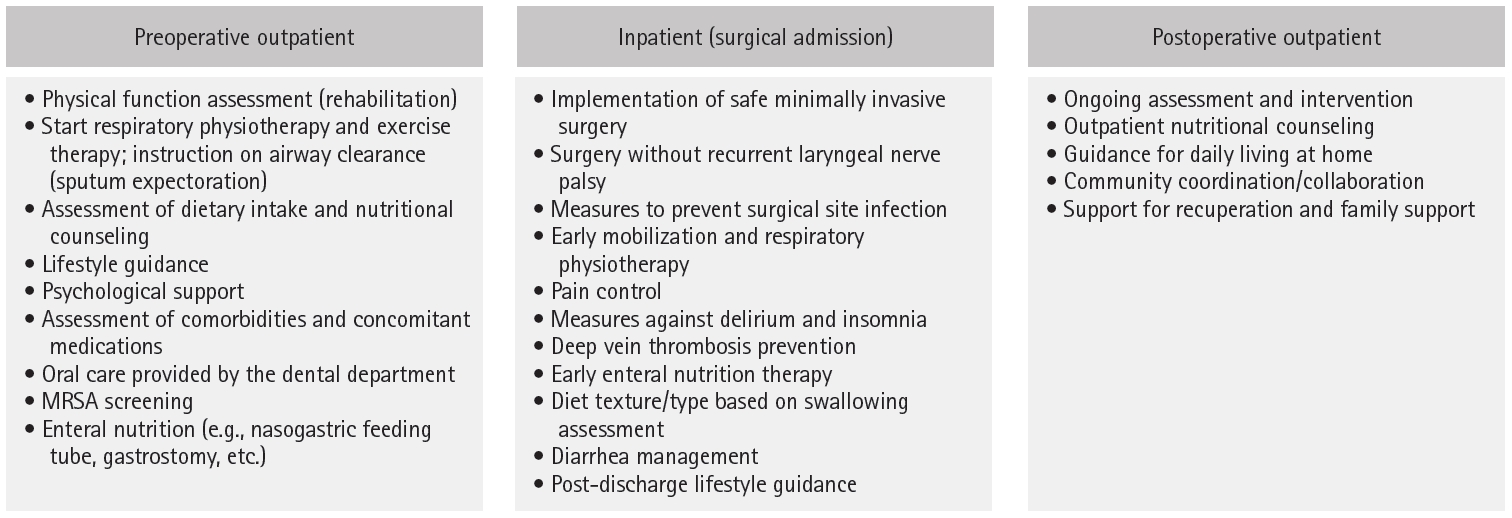
| HOPE | Current study (2017) | Physical fitness measurement (rehabilitation), respiratory/physical therapy, sputum discharge guidance, dietary intake assessment/nutritional guidance, lifestyle guidance, psychological care, comorbidity/medication assessment, oral care by dental department, MRSA screening, enteral nutrition (nasal feeding tube, gastrostomy, etc.) | Practice of safe minimally invasive surgery, surgery without recurrent laryngeal nerve palsy, SSI prevention, early mobilization/respiratory physiotherapy, pain control, delirium/insomnia prevention, deep vein thrombosis prevention, early enteral nutrition therapy, dietary form based on swallowing evaluation, management of diarrhea, post-discharge lifestyle guidance | Reduced incidence of atrial fibrillation, reduced pneumonia, increased skeletal muscle mass |
- 1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A, et al. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87-108. ArticlePubMedPDF
- 2. Weimann A, Braga M, Harsanyi L, Laviano A, Ljungqvist O, Soeters P, et al. Espen guidelines on enteral nutrition: surgery including organ transplantation. Clin Nutr 2006;25:224-44. ArticlePubMed
- 3. Shirakawa Y, Tanabe S, Fujiwara T. Forefront of perioperative team medicine for esophageal cancer surgery. Nihon Shokakibyo Gakkai Zasshi 2019;116:138-44. ArticlePubMed
- 4. Shirakawa Y, Noma K, Maeda N, Tanabe S, Sakurama K, Sonoyama-Hanaoka A, et al. Early intervention of the perioperative multidisciplinary team approach decreases the adverse events during neoadjuvant chemotherapy for esophageal cancer patients. Esophagus 2021;18:797-805. ArticlePubMedPDF
- 5. Watanabe M, Mine S, Nishida K, Yamada K, Shigaki H, Oya S, et al. Improvement in short-term outcomes after esophagectomy with a multidisciplinary perioperative care team. Esophagus 2016;13:337-42. ArticlePDF
- 6. Ochiai R. The case of Toho University Omori Medical Center Hospital. Jpn J Clin Anaesthesiol 2018;42:1190-8.
- 7. Yamamoto C, Sakakibara S. The evolution of preoperative outpatient care: looking back on the 10 years since its establishment and considering the future of preoperative outpatient care. J Jpn Soc Surg 2023;44:267-71.
- 8. Yokoyama K. Establishment of a perioperative management team in a regional hospital and initiatives for pre- and postoperative swallowing function assessment. J Jpn Soc Surg 2021;42:221-4.
- 9. Ishida T. Perioperative nutritional management by TOPS (Tobu Hospital Patient Support Center). J Jpn Soc Surg 2023;44:129-32.
- 10. Takeuchi H, Ikeuchi S, Kawaguchi Y, Kitagawa Y, Isobe Y, Kubochi K, et al. Clinical significance of perioperative immunonutrition for patients with esophageal cancer. World J Surg 2007;31:2160-7. ArticlePubMedPDF
- 11. Japanese Society for Parenteral and Enteral Nutrition. Guidelines for Parenteral and Enteral Nutrition, 3rd edition [Internet]. Japanese Society for Parenteral and Enteral Nutrition; 2014. [cited 2023 Jan 1]. Available from: https://files.jspen.or.jp/2014/04/201404QR_guideline.pdf
- 12. Haneda R, Hiramatsu Y, Kawata S, Soneda W, Booka E, Murakami T, et al. Clinical impact of diarrhea during enteral feeding after esophagectomy. Int J Clin Oncol 2024;29:36-46. Article
- 13. Sato H, Miyawaki Y, Fujiwara N, Sakuramoto S, Okamoto M, Yamaguchi S, et al. Exercise therapy in esophageal surgery. Surg Metab Nutr 2018;52:289-93.ArticlePubMedPDF
- 14. Takesue T, Takeuchi H, Ogura M, Fukuda K, Nakamura R, Takahashi T, et al. A prospective randomized trial of enteral nutrition after thoracoscopic esophagectomy for esophageal cancer. Ann Surg Oncol 2015;22(Suppl 3):S802-9. ArticlePubMedPDF
- 15. Kawata S, Hiramatsu Y, Shirai Y, Watanabe K, Nagafusa T, Matsumoto T, et al. Multidisciplinary team management for prevention of pneumonia and long-term weight loss after esophagectomy: a single-center retrospective study. Esophagus 2020;17:270-8. ArticlePubMedPMCPDF
- 16. Honke J, Hiramatsu Y, Kawata S, Booka E, Matsumoto T, Morita Y, et al. Usefulness of wearable fitness tracking devices in patients undergoing esophagectomy. Esophagus 2022;19:260-8. ArticlePubMedPDF
References
Figure & Data
REFERENCES
Citations

- Figure
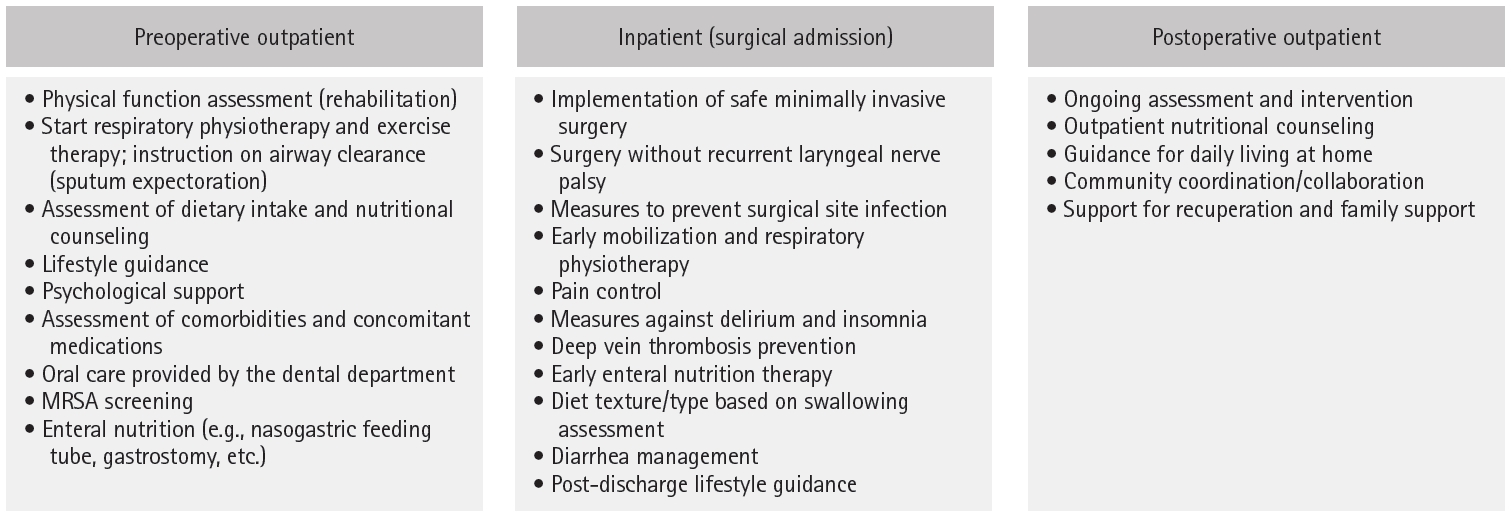
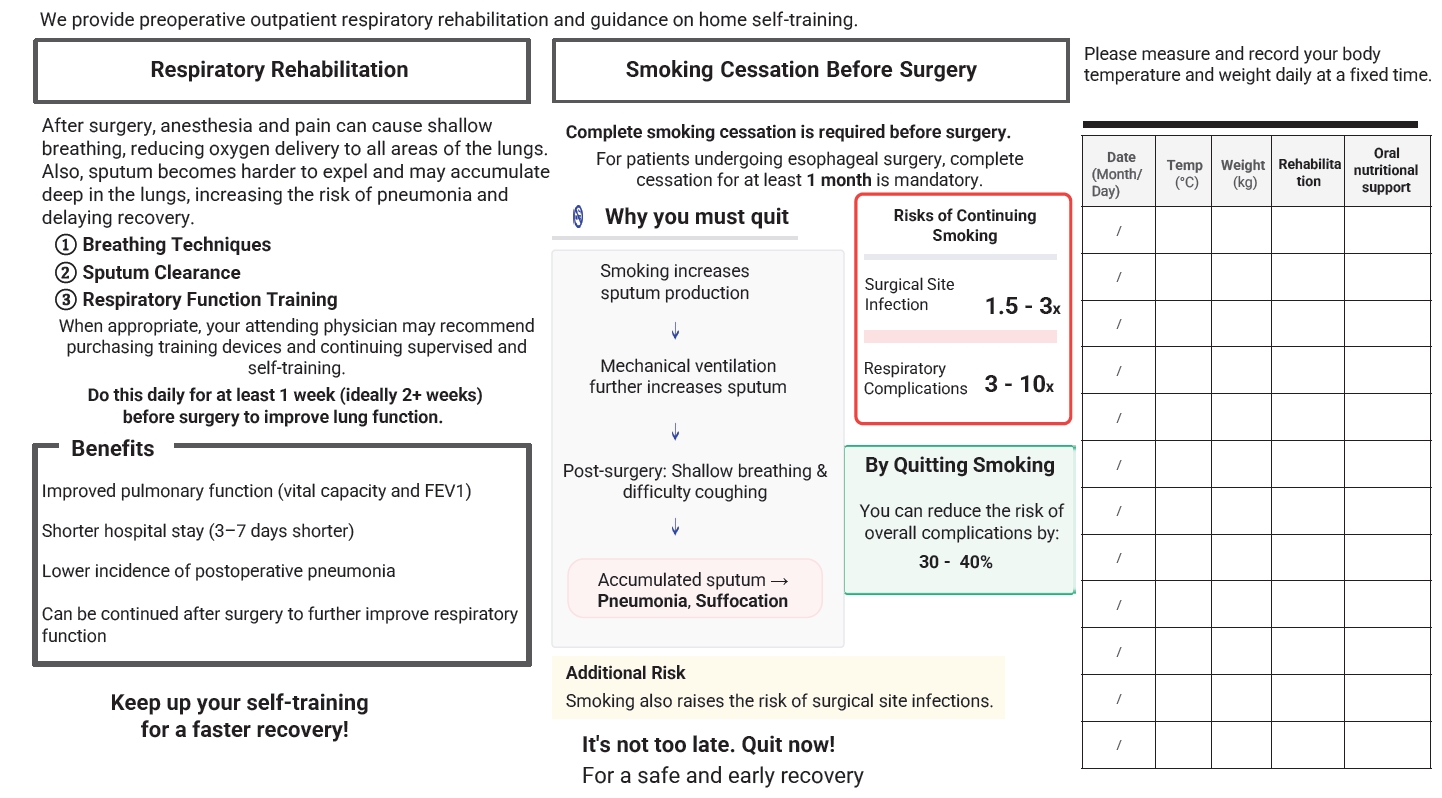
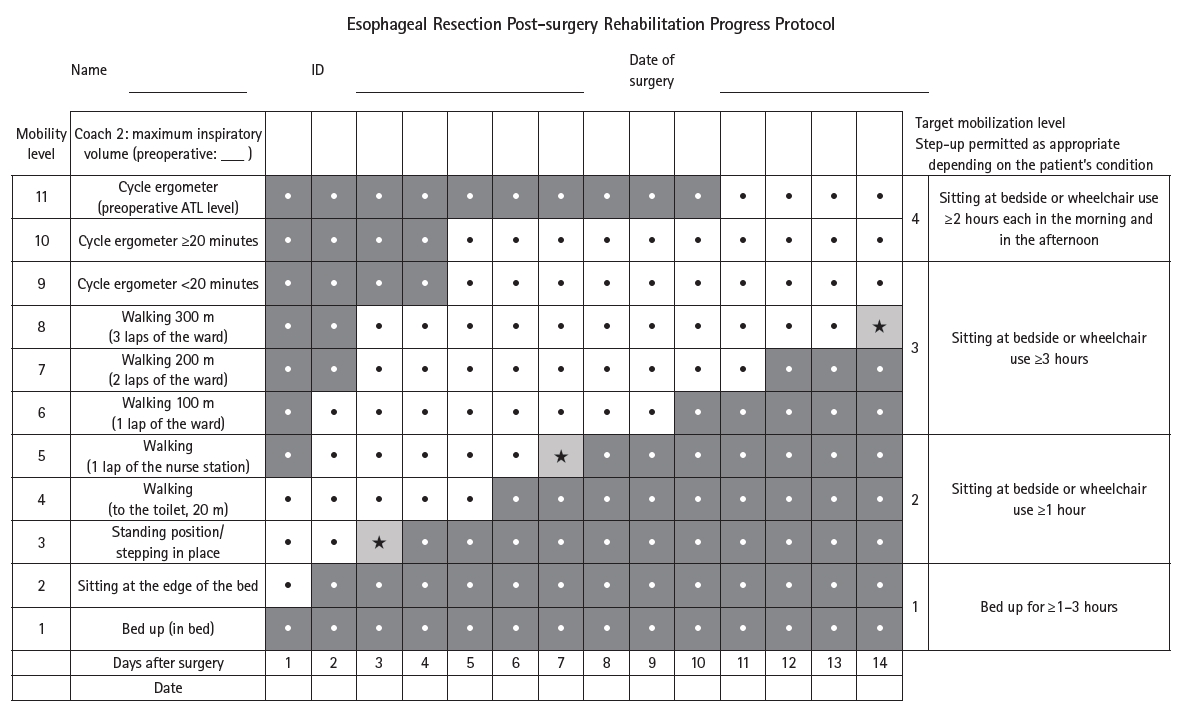
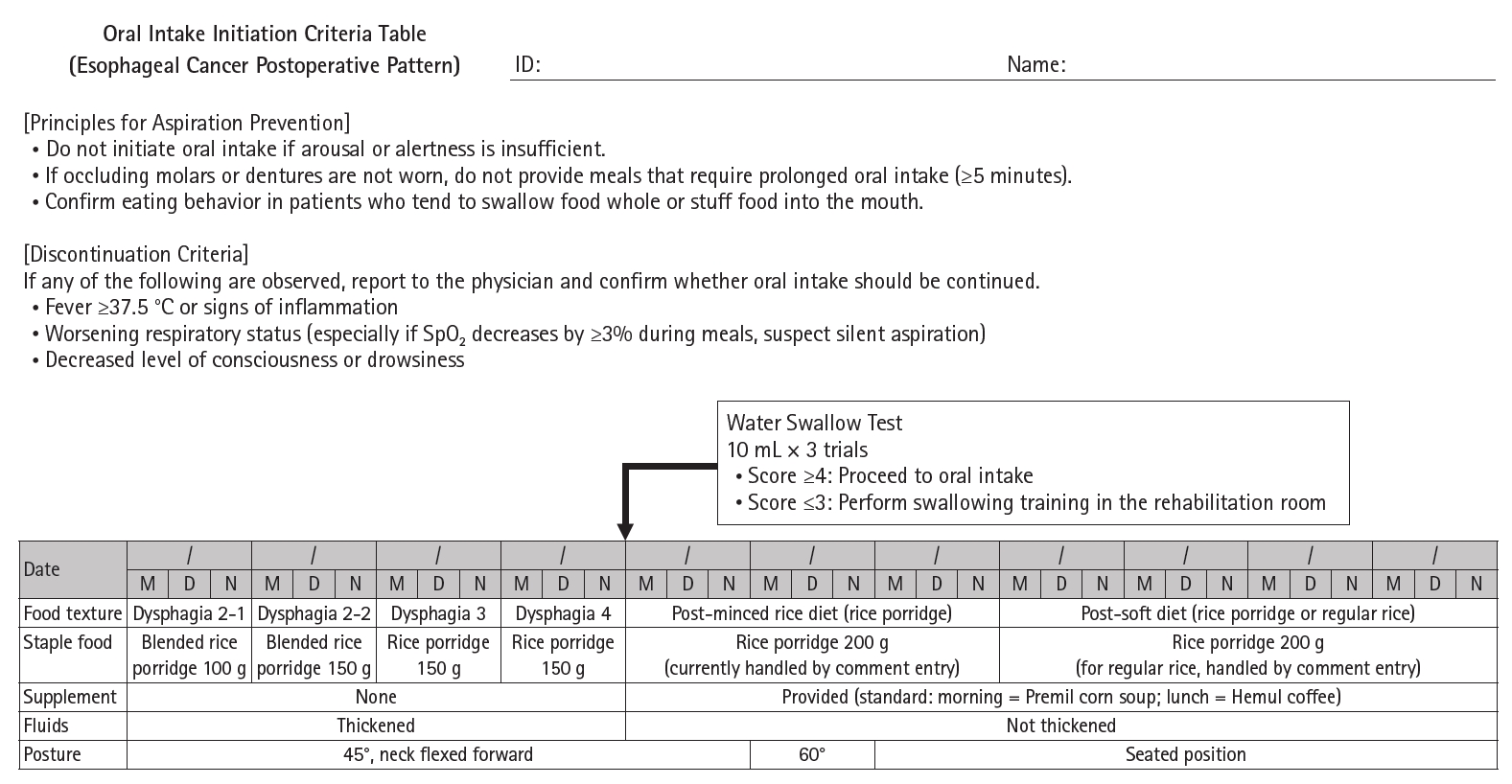
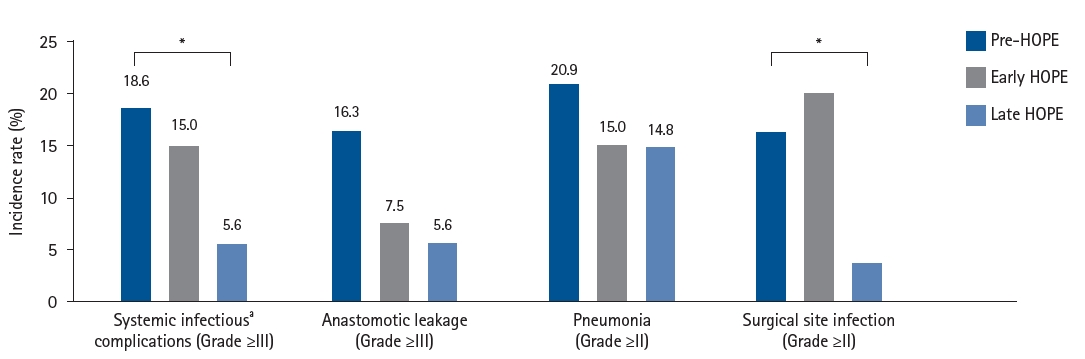
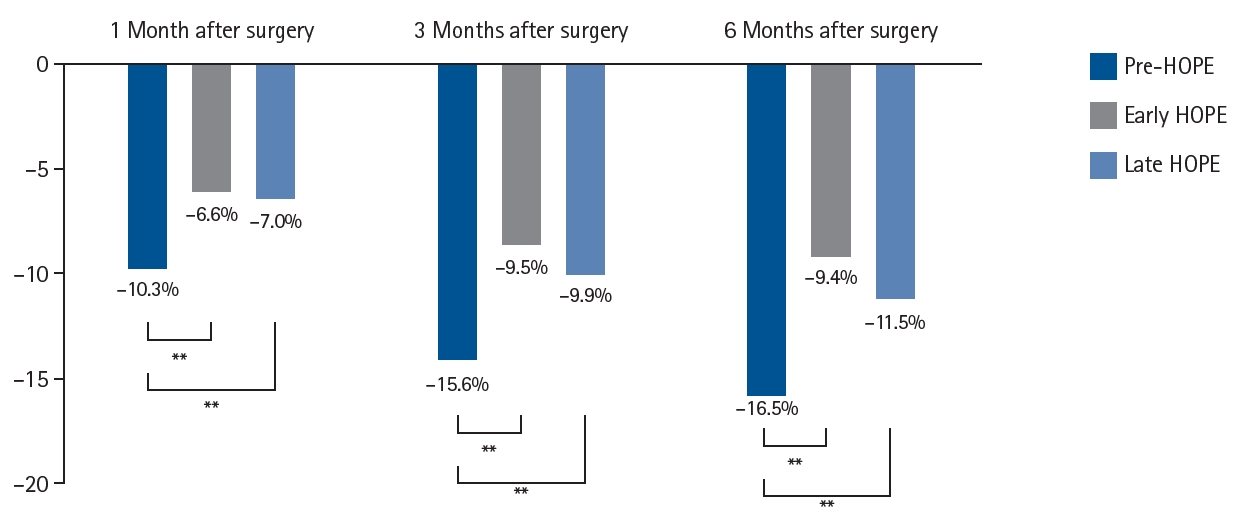
Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Fig. 5.
Fig. 6.
| Perioperative management team | Author | Preoperative | Postoperative | Outcome |
|---|---|---|---|---|
| PERiO | Shirakawa et al. (2019) [3] | Smoking cessation guidance, physical ability assessment, exercise therapy, oral care | Physical ability assessment, respiratory physiotherapy, basic movement assistance, eating/swallowing rehabilitation | Fewer days to postoperative ambulation, shortened hospital stay, reduced adverse events during preoperative chemotherapy |
| Shirakawa et al. (2021) [4] | ||||
| PeriCan | Watanabe et al. (2016) [5] | Alcohol and smoking cessation guidance, oral care | Early mobilization, rehabilitation, swallowing function assessment, dietary guidance | Reduced pneumonia |
| Perioperative Center | Ochiai (2018) [6] | Patient orientation, oral care, medication adjustment, deep vein thrombosis prevention | Postoperative pain control, oral care | Shortened average hospital stay |
| PMT | Yamamoto and Sakakibara (2023) [7] | Smoking/alcohol cessation guidance, respiratory rehabilitation, nutritional guidance, oral care, skin protection, consultation for use of economic/social resources (post-discharge life support, nursing care, etc.) | Continued postoperative nursing, discharge support, utilization of social resources | Reduced surgery postponement, optimized hospital stays, prevention of complications |
| PMT | Yokoyama (2021) [8] | Physical and social background, patient/family acceptance of surgery, swallowing function assessment, risk of postoperative nausea/vomiting, preoperative pain management plan | Treatment course, evaluation of future PMT interventions, coordination with each committee | Identification of high-risk patients for swallowing disorders |
| Tobu Hospital Patient Support Center | Ishida (2023) [9] | Assessment of dietary intake, nutritional guidance, grip strength/body measurement, calculation of nutritional indices, body composition measurement, prehabilitation (aerobic exercise, resistance exercise, stretching, respiratory training), oral care | Nutritional management, rehabilitation | Increased walking distance, improved sit-and-reach, increased skeletal muscle mass |
| HOPE | Current study (2017) | Physical fitness measurement (rehabilitation), respiratory/physical therapy, sputum discharge guidance, dietary intake assessment/nutritional guidance, lifestyle guidance, psychological care, comorbidity/medication assessment, oral care by dental department, MRSA screening, enteral nutrition (nasal feeding tube, gastrostomy, etc.) | Practice of safe minimally invasive surgery, surgery without recurrent laryngeal nerve palsy, SSI prevention, early mobilization/respiratory physiotherapy, pain control, delirium/insomnia prevention, deep vein thrombosis prevention, early enteral nutrition therapy, dietary form based on swallowing evaluation, management of diarrhea, post-discharge lifestyle guidance | Reduced incidence of atrial fibrillation, reduced pneumonia, increased skeletal muscle mass |
| WFT use (n=31) | No WFT use (n=31) | P-value | |
|---|---|---|---|
| All complications, No. (%) | |||
| C-D grade II or higher | 5 (16.1) | 12 (38.7) | 0.043 |
| Anastomotic leakage | 3 (9.7) | 2 (6.5) | 0.500 |
| Pneumonia | 0 | 7 (22.6) | 0.005 |
| Postoperative hospital stay (day), median (range) | 22 (20–29) | 29 (24–36) | 0.012 |
| Blood test results at 1 month after surgery, median (range) | |||
| Prognostic nutritional index | 46.2 (40.8–49.7) | 42.6 (37.8–45.9) | 0.034 |
| Albumin (g/dL) | 3.9 (3.6–4.1) | 3.6 (3.2–3.9) | 0.013 |
| Transthyretin (mg/dL) | 24.4 (21.5–26.0) | 19.4 (15.0–22.9) | 0.001 |
| CRP (mg/dL) | 0.14 (0.09–0.31) | 0.35 (0.15–1.60) | 0.018 |
PERiO, Perioperative Management Center; PeriCan, Perioperative Team at the Cancer Institute Hospital; PMT, Perioperative Management Team; MRSA, methicillin-resistant
WFTs, wearable fitness tracking devices; C-D, Clavien-Dindo classification; CRP, C-reactive protein.

 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN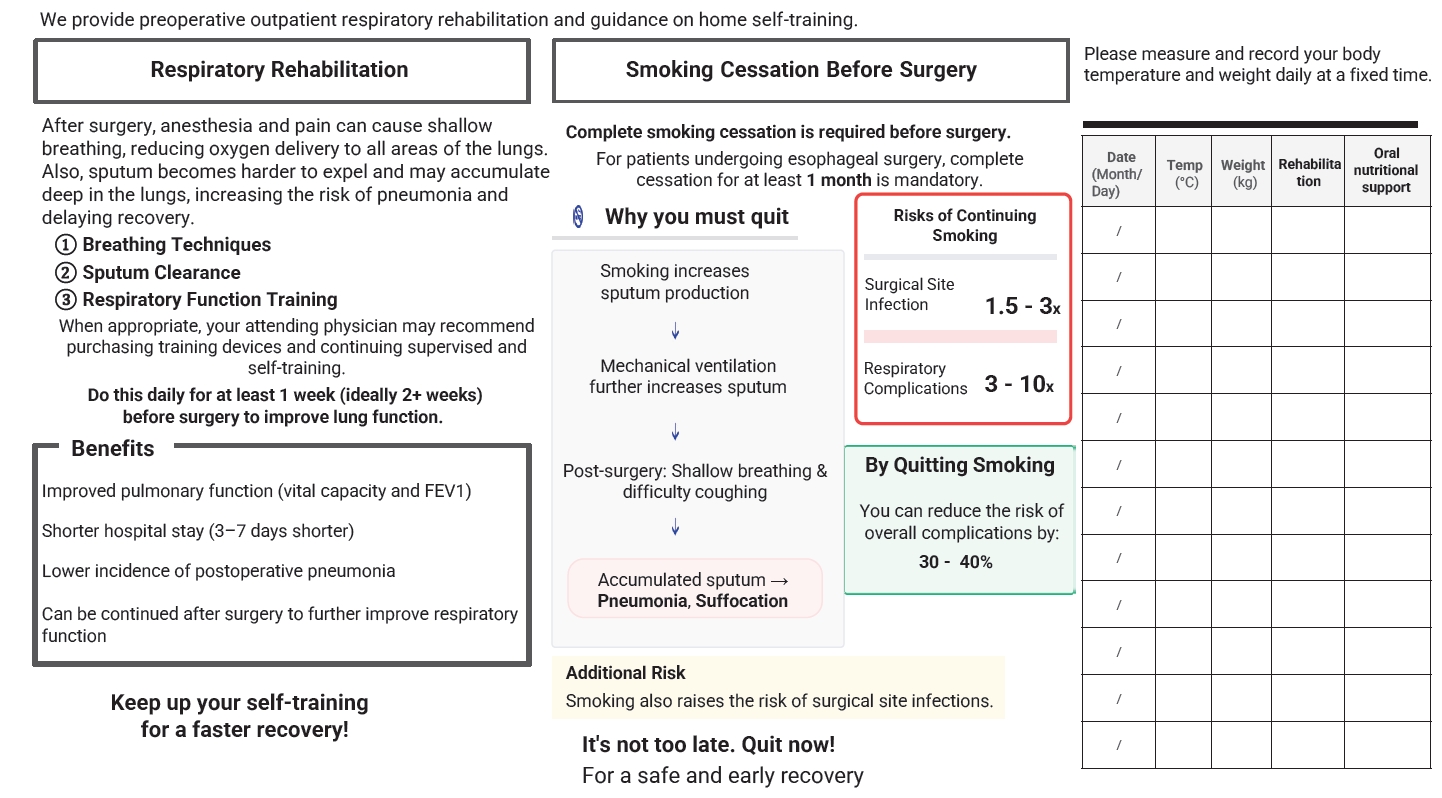
 Cite
Cite